Abstract
Introduction: Cytogenetic abnormalities are an important prognostic factor for predicting overall survival (OS) and relapse incidence (RI) in children with acute myeloid leukemia (AML). Although allogeneic hematopoietic cell transplant (HCT) is generally recommended for patients with AML in first complete remission (CR1) and poor-risk (PR) cytogenetics, whether these cytogenetic and molecular features retain their prognostic value after HCT is unknown. Additionally, PR AML is a heterogeneous category that includes several distinct cytogenetic abnormalities, some of which might have a worse prognosis than others. In this study, we evaluated whether the cytogenetic risk classification at diagnosis of AML was predictive of post-HCT outcomes in pediatric patients.
Methods: All patients <18 years of age at the time of HCT reported to the European Society for Blood and Marrow Transplantation (EBMT) registry, who had received their first allogeneic HCT for AML in CR1 between 2005 and 2020, and who had their karyotype evaluation at diagnosis available were included in this analysis. Patients were subgrouped as (a) monosomy 7/del7q or monosomy 5/del5q, (b) 11q23 abnormality excluding t(9;11), (c) complex or monosomal karyotype, or (d) other. A complex karyotype was defined as one with three or more structural abnormalities, and a monosomal karyotype was defined as a monosomy with one or more structural abnormalities, excluding WHO-designated recurring translocations or inversions based on 2017 ELN recommendations. Other cytogenetic abnormality subgroup included t(6;9), t(3;5), t(9;22), t(8;16), inv(3) or t(3;3), t(16;21), abn(11p15), and del(12p) or abn(12p13). Cytogenetic subgroup, age at HCT, whether HCT was from a female donor to a male recipient, patient/donor CMV status, time from AML diagnosis to HCT, and year of HCT were included as predictors for OS, RI and non-relapse mortality (NRM) in the multivariate analysis.
Results: We included 744 pediatric patients (42.2% female; median age at HCT: 8.6 years [range: 0.3-18 years]) from 139 participating centers in the study. Median follow-up after HCT was 4.4 years. Thirty-seven percent had an 11q23 abnormality, 24% had monosomy 7/del7q or monosomy 5/del5q, 24% had a complex or monosomal karyotype, and 15% met the criteria for other PR cytogenetic abnormalities. Only 7% had secondary AML. Pre-HCT minimal residual disease (MRD) data were available for 346 patients, 86% of whom were MRD negative at the time of HCT. Patients received grafts from a matched related donor (27%), a mismatched related donor (11%), an unrelated donor (46%), or a cord blood donor (16%). Of the total patients, 55% received bone marrow, 29% received peripheral blood-derived stem cells and the rest (16%) received cord blood as the stem cell source. Myeloablative conditioning was used in 97% of the patients.
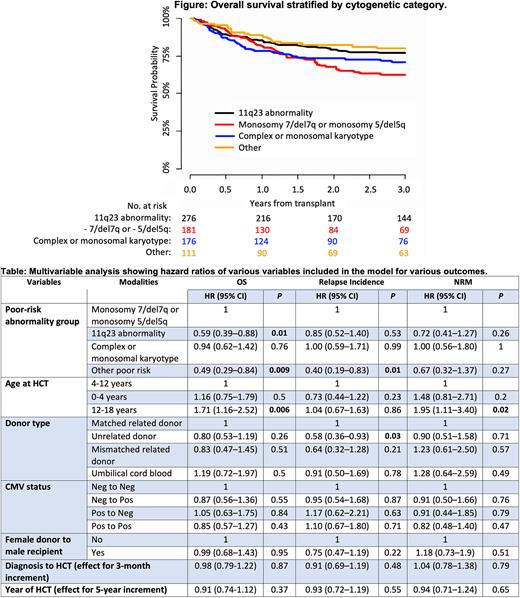
The OS and leukemia free survival for the entire cohort was 76% and 70% respectively at 2 years. In a multivariate model, 11q23 (hazard ratio [HR] = 0.59, P = 0.01) and other poor-risk cytogenetic abnormalities (HR = 0.49, P < 0.01) were associated with significantly better OS when compared with monosomy 7/del7q or monosomy 5/del5q. The "other PR cytogenetic abnormalities" category was also associated with a lower risk of disease relapse after HCT (HR = 0.4, P = 0.01). Receiving an HCT from an unrelated donor, as compared with a matched related donor, was associated with lower RI (HR = 0.58, P = 0.03). Additionally, older age at HCT (12-18 years) was associated with the highest risk of NRM (HR = 1.95, P = 0.02) and with worse OS (HR = 1.71, P < 0.01) than seen in younger patients (aged 4-12 years).
Conclusion: PR cytogenetic abnormalities at diagnosis remain predictive of OS after HCT for AML in pediatric patients. Monosomy 7/del7q or monosomy 5/del5q confer a poor prognosis even after HCT, whereas 11q23 abnormality and other PR cytogenetic abnormalities predict a favorable outcome after HCT. Our data also suggest that an HCT from an unrelated donor may offer greater protection against relapse than an HCT from a matched related donor, but this requires further validation. Efforts for improving conditioning regimens and transplant techniques should focus on reducing NRM and reducing RI in this population.
Disclosures
Sharma:Vindico Medical Education: Honoraria; CRISPR Therapeutics: Research Funding; Medexus Inc: Consultancy; Spotlight Therapeutics: Consultancy; Magenta Therapeutics: Other: Research collaboration; Novartis: Other: Other; Vertex Pharmaceuticals/CRISPR Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Other. Locatelli:BlueBird bio: Speakers Bureau; Miltenyi: Speakers Bureau; Medac: Speakers Bureau; Novartis: Honoraria, Speakers Bureau; Amgen: Speakers Bureau; SOBI: Speakers Bureau; Jazz Pharmaceuticals: Honoraria; Neovii: Speakers Bureau. Fagioli:AMGEN: Membership on an entity's Board of Directors or advisory committees, Research Funding; JAZZ PHARMA: Membership on an entity's Board of Directors or advisory committees, Research Funding; NOVARTIS: Membership on an entity's Board of Directors or advisory committees, Research Funding; CLINIGEN: Membership on an entity's Board of Directors or advisory committees, Research Funding; TAKEDA: Membership on an entity's Board of Directors or advisory committees, Research Funding; GENZYME SANOFI: Membership on an entity's Board of Directors or advisory committees, Research Funding; PFIZER: Membership on an entity's Board of Directors or advisory committees, Research Funding. Biffi:Altheia Science: Consultancy. Tambaro:Gilead: Speakers Bureau; Jazz: Other: Meeting Participation Fees, Speakers Bureau; Novartis: Other: Meeting Participation fes. Bhatt:Rite Aid Corp.: Divested equity in a private or publicly-traded company in the past 24 months; Johnson & Johnson: Divested equity in a private or publicly-traded company in the past 24 months; Moderna Inc.: Divested equity in a private or publicly-traded company in the past 24 months; Pfizer Inc.: Divested equity in a private or publicly-traded company in the past 24 months.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal